We back your science.
We help bring it to life.
We give you what you need to go further.
If you're advancing clinical-stage research for a rare disease, we help shape it into a compelling product candidate and connect it with pharma to bring it closer to patients.
Our platform is made for people and organizations actively working on clinical-stage research for rare diseases, including Universities, Professors, Research Centers, Tech Transfer Offices, Hospitals, Principal Investigators, Biotech Labs, and Government Labs.
What we'll accomplish together
Transform your research into a market-ready medical product that attracts pharmaceutical partners
You Provide
- Core project data and clinical research documentation
- Scientific input and iterative feedback
We Transform It Into
- A well-defined medical product candidate, potentially enriched with complementary research
- A complete R&D and regulatory strategy, with timelines and milestones
- Financial models tailored to clinical and commercial potential
- IP protection and market exclusivity
- Manufacturing strategy and access to qualified partners
- Direct connections with pharmaceutical companies and foundations
Together, We Reach Patients
You collaborate with the pharma partner to bring your therapy to market through:
How it works
A comprehensive process designed for people and organizations advancing rare disease therapies
This process is specifically designed for people and organizations working on clinical trials that test better or new therapies for rare diseases.
If your disease isn't listed but you know it's quite rare, register with us and we'll help search for it.
Register & Explain Your Research
Create your account and provide comprehensive details about your rare disease research project. Tell us about your clinical trial, the targeted disease, your research methodology, and evidence that supports your claims.
- Research background and objectives
- Clinical trial design and methodology
- Target rare disease and patient population
- Current development stage
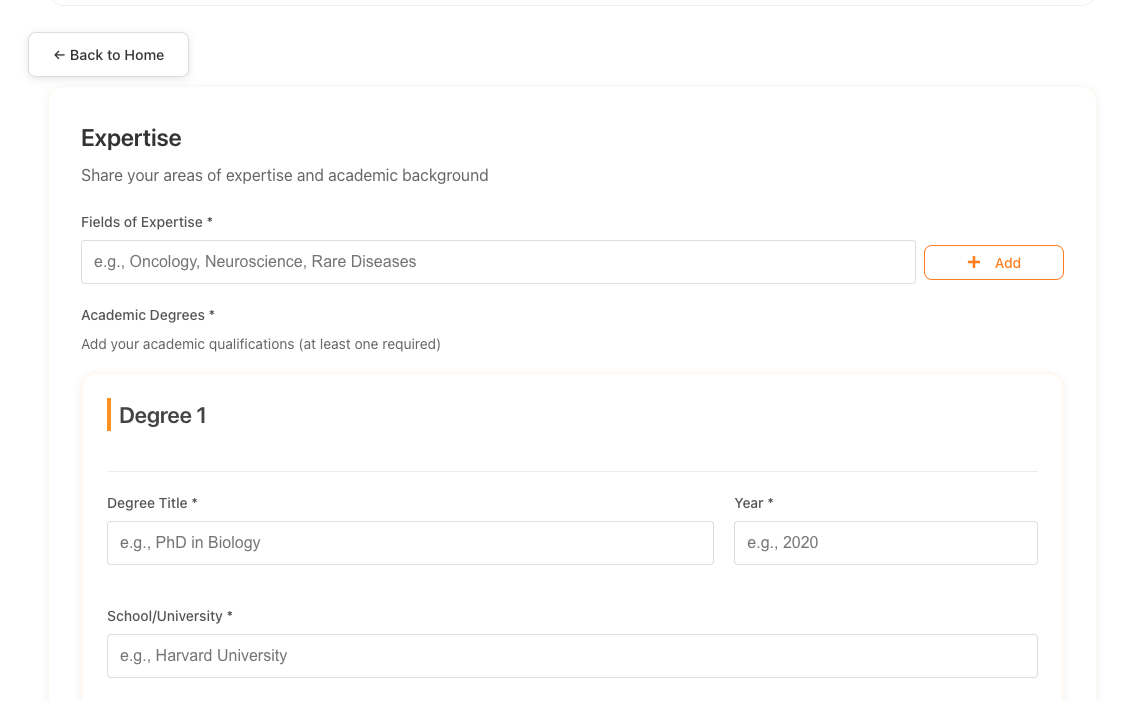
Registration Platform Screenshot
Upload Research Files
You'll upload all relevant research files and documentation. Our platform will guide you on what to include and what NOT to upload for security and compliance reasons.
- Clinical trial protocols and documentation
- Regulatory documentation
- Research publications and IP information
- Safety and efficacy data and publications
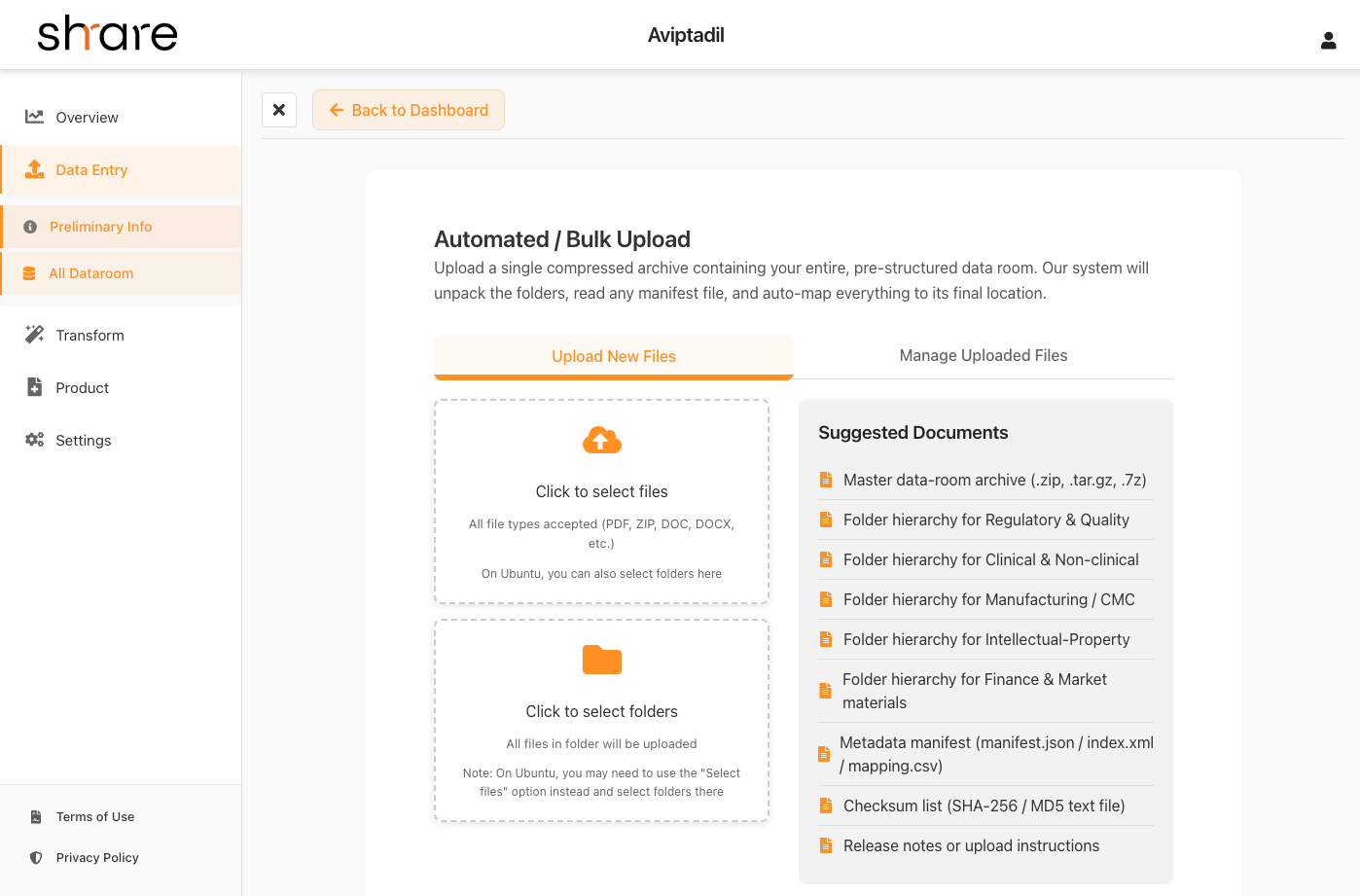
File Upload Interface Screenshot
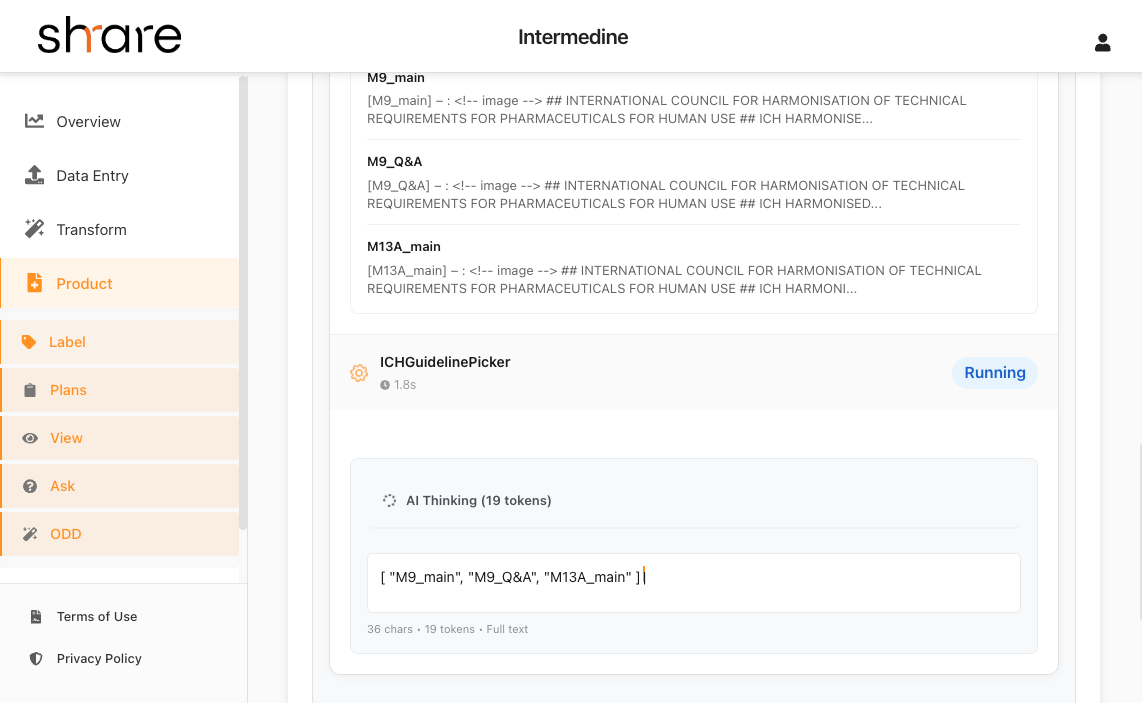
Data Processing Dashboard Screenshot
Incubation
Our AI-powered incubation process enriches your research data with global clinical databases, regulatory intelligence, market insights and combining it with other research in our community if needed. Working collaboratively with AI and human expertise, we transform your raw research into a comprehensive, market-ready medical product candidate.
- Data enrichment with complementary research and global clinical evidence
- eCTD documentation and automated regulatory submissions
- Complete R&D strategy with success probabilities and optimized timelines
- Financial modeling for development costs and market projections
- IP, market exclusivity and manufacturing partner identification
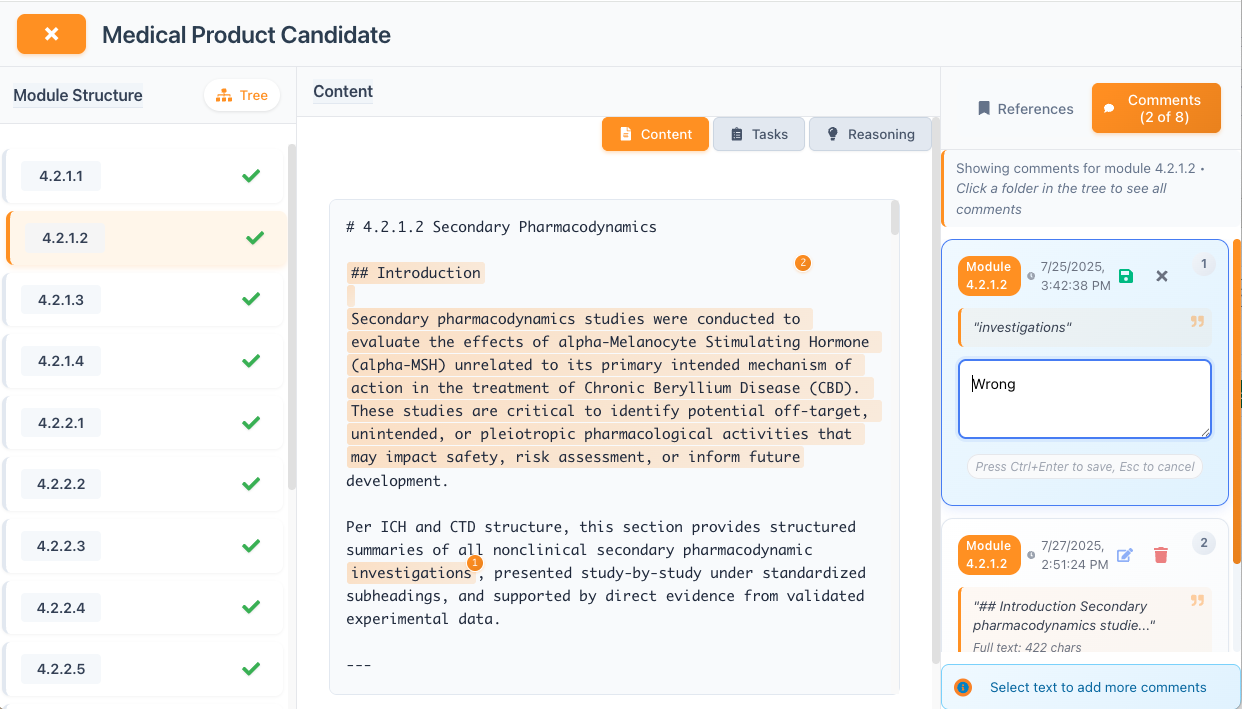
Expert Collaboration & Feedback
We value your scientific expertise! Shrare will periodically request your professional judgment and feedback on the work we're producing in your area of expertise. This collaborative approach ensures accuracy and leverages your deep knowledge while requiring minimal time commitment.
- Review AI-generated summaries and analyses
- Provide expert feedback on technical aspects
- Validate scientific interpretations
- Time commitment: Maximum 2 hours per week
Collaboration Interface Screenshot
Partner Matching Dashboard Screenshot
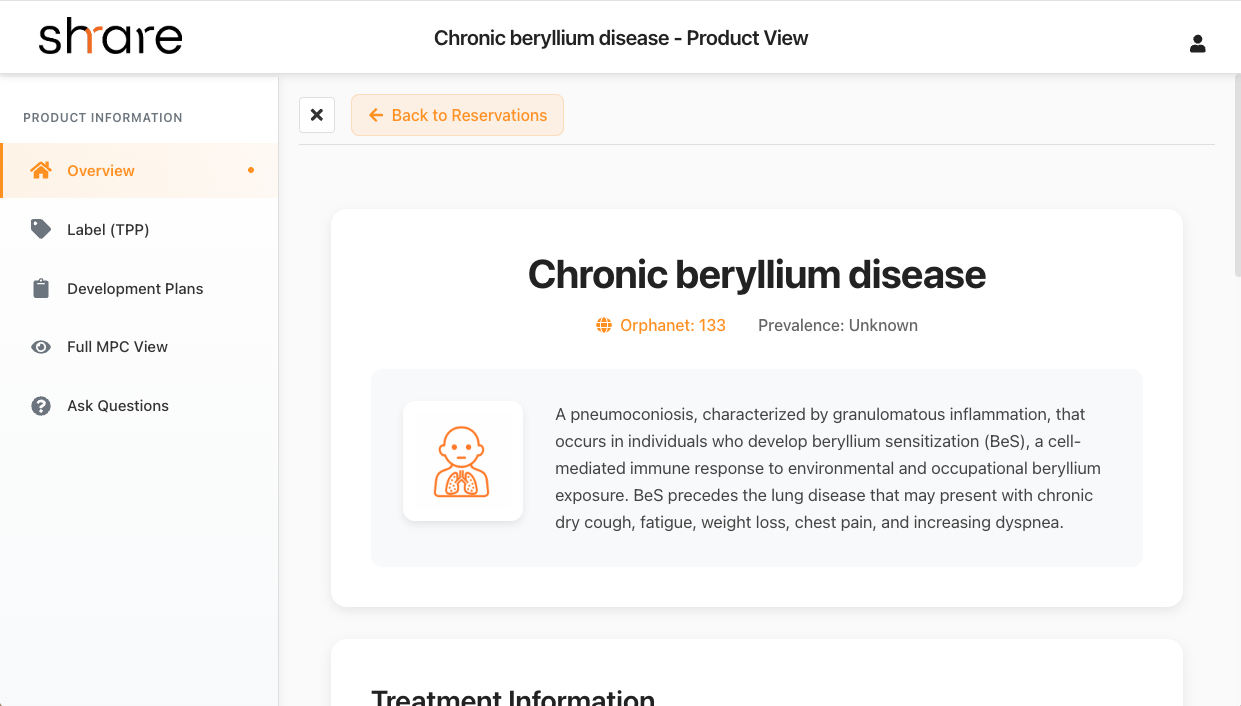
Partner Matching & Market Execution
Once all is ready, we focus on finding a pharmaceutical company, foundation, or partner that wants to bring your therapy forward. Shrare actively seeks the right partners to bring your therapy to market through our comprehensive network.
- Matching with pharmaceutical companies interested in rare diseases
- Connecting with foundations focused on your therapeutic area
- Identifying strategic partners for development and commercialization
- Facilitating partnerships that maximize therapeutic impact
- Supporting ongoing collaboration through development phases
Key Benefits for Researchers
Comprehensive support to maximize your research impact and value
Regulatory & Commercial Strategy
We prepare for you the R&D and regulatory strategy, with required documentation and interactions with regulators, and help position your project for maximum market impact.
Start my journeyFinancial & Cost Efficiency
Unlock the full financial potential of your research with structured deals, milestone payments, and reduced development costs.
Unlock my potentialGlobal Network Access
Tap into a curated global network of pharma companies, biotech firms, and investors ready to support clinical-stage innovation.
Join the platformConsulting & Clinical Continuity
Pharma sponsors may request paid consultancy, scientific collaboration, and co-development of trials—amplifying your visibility and extending your research journey.
Grow with SHRARESpeed & Efficiency
Cut down the time and cost of reaching therapy, thanks to automated processes and a ready-to-collaborate ecosystem.
Accelerate NowImpact on Patients
Contribute directly to bringing new treatments to millions of rare-disease patients who currently have no therapeutic options.
Make a DifferenceFrequently Asked Questions
Everything you need to know about partnering with Shrare
Breakthroughs deserve momentum. SHRARE removes barriers and accelerates your path to patients—offering regulatory guidance, funding opportunities, and direct access to pharma partners.
We help build long-term alliances between researchers and pharma—when possible, even beyond licensing—so you can focus on what you do best: science.
Researchers benefit from multiple revenue streams and cost savings:
- Upfront payments upon licensing
- Milestone-based payments tied to development progress
- Royalties from market success
- Lower costs through our streamlined SHRARE Incubator with human-in-the-loop support
Pharma partners may invite researchers to:
- Join scientific advisory boards
- Co-author publications and present at major conferences
- Co-lead clinical trials and further development phases
These collaborations amplify your visibility and extend your research journey beyond the initial licensing.
With operations in the U.S., Switzerland, and the UAE, SHRARE offers your research global visibility and access to diverse international partners.
We streamline your journey by:
- Accelerating regulatory approvals
- Providing structured financial support
- Matching you with the right pharma partners using AI
- Managing commercialization logistics so you can focus on science
Institutions benefit from:
- New funding streams through licensing and royalties
- Increased prestige via high-impact partnerships
- IP protection and global commercialization
- Expanded industry relationships through our platform ecosystem
Ready to unlock the full potential of your research?
Don't let your project remain on a shelf. Join researchers around the world using SHRARE to turn clinical-stage science into real-world therapies.
Start Your Journey